Clinical case simulator “Pregnancy after myocardial revascularization”. A young woman who has had multiple recurrent myocardial infarctions and coronary artery stenting learns about her pregnancy and decides to keep the pregnancy. What are the features of managing such a group of patients? Is the correction of drug therapy necessary? What can be the tactics of patient management, taking into account the planned delivery?
- You will communicate with the patient in dialogue simulators;
- Will conduct an objective examination of the patient;
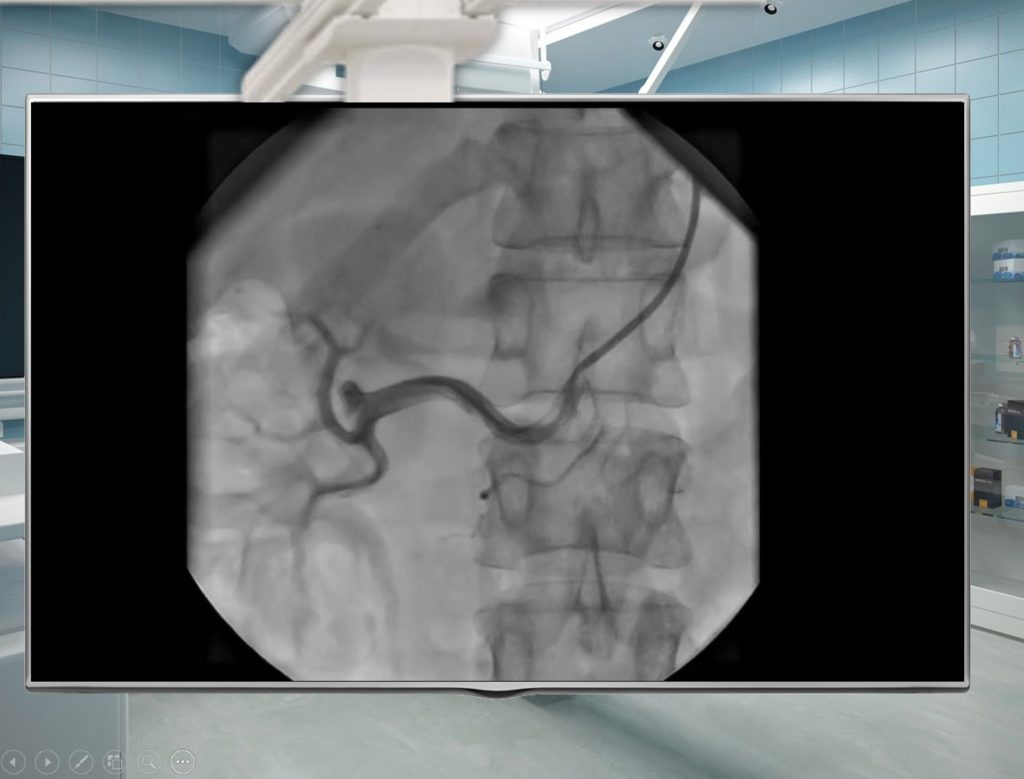
- Will reexamine patient’s СD’s with imaging studies;
- Will carry out the necessary additional examinations;
- You will take part in the obstetric and cardiological consultation;
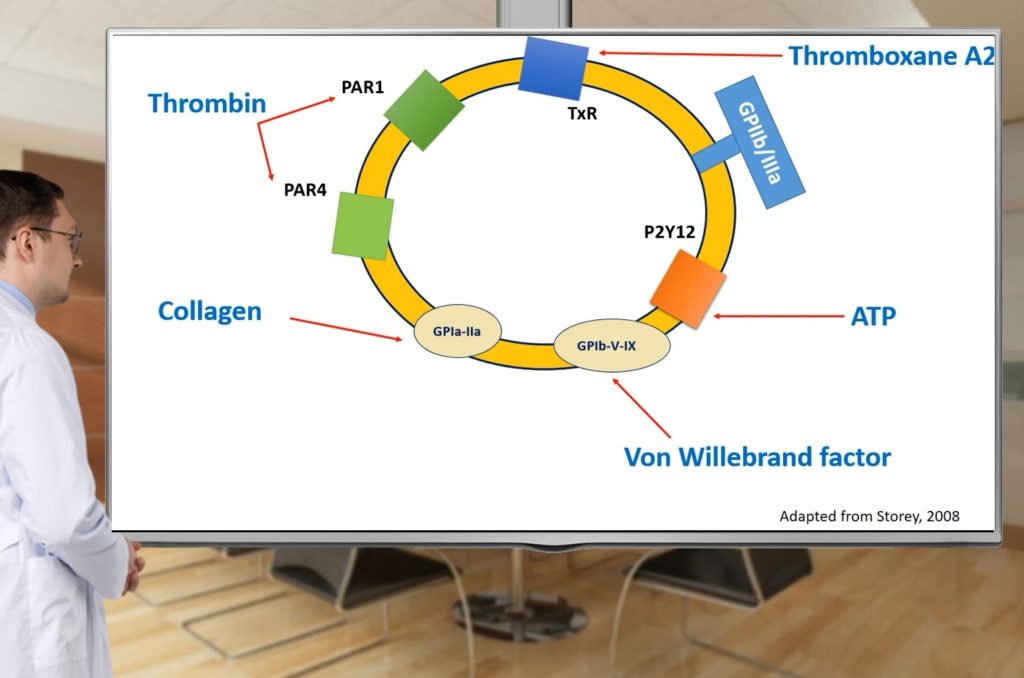
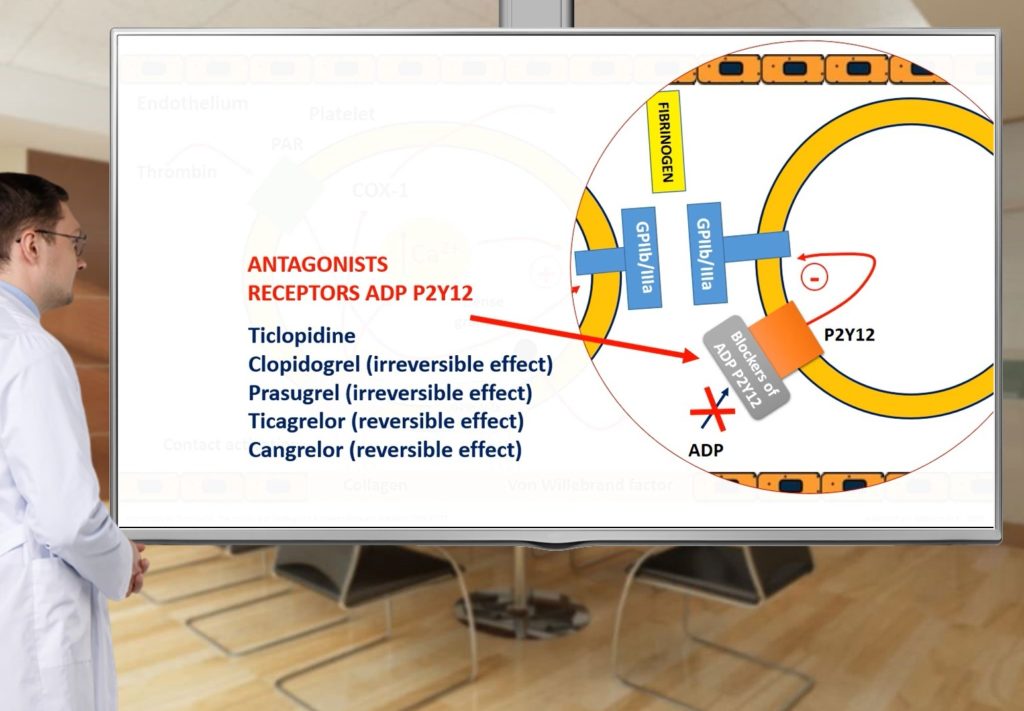
- Will remember structure and function of platelets;
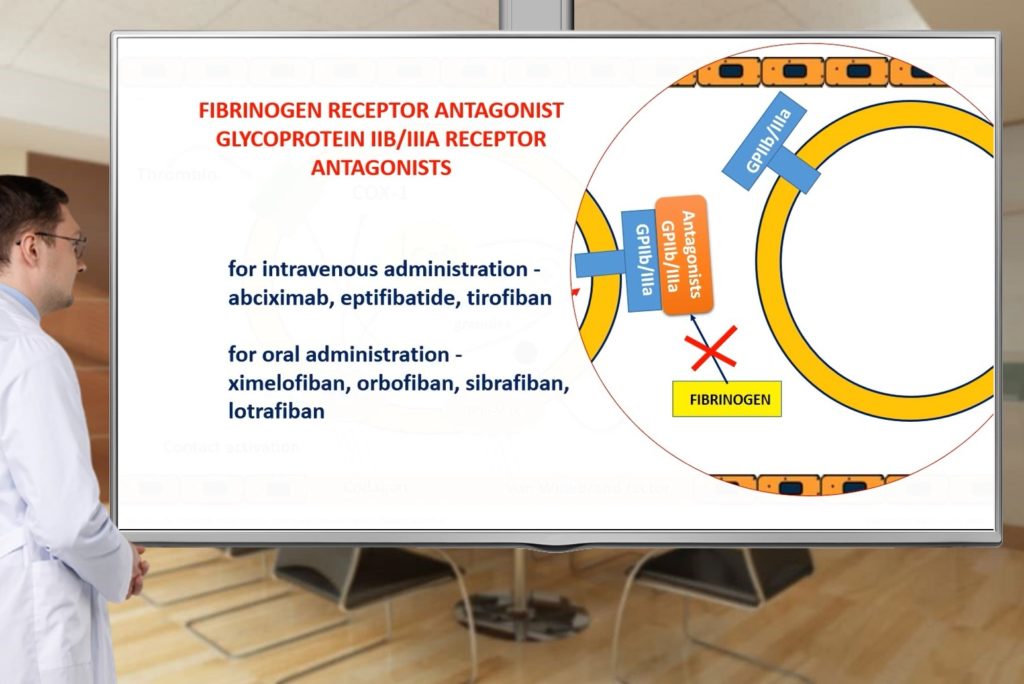
- Will remember classification of antiplatelet drugs;
- Will analyze types of hypertension in pregnant women;
- Will get acquainted with the classification of drugs by hazard class in pregnant women;
- Will get acquainted with the general algorithm of drug therapy for patients after myocardial revascularization in the context of planning for non-cardiac surgery;
- Will сhoose the most optimal drug therapy strategy for the patient, taking into account the pregnancy and the upcoming delivery.
Clinical case simulator “Pregnancy after myocardial revascularization”:
- More than 9 locations;
- 4 interactive simulators;
- Consilium;
- Gamification;
- Branching-scenario;
- Visualization of diagnostic tests;
- Over 90 practice questions with explanations and comments;
- Analysis of clinical guidelines based on a particular clinical situation;
- Number of competencies – 18.
Total duration: 2–2.5 hours.
Devices for training in the clinical case simulator:
- Tablets
- Laptops
- Computers
One-time purchase of one simulator
Sign upClinical case
Feedback
Summing up
Recommended literature
Сase simulator discussion
Associated case simulator: “Choosing the duration of DAPT administration after coronary stenting in a young woman”























Responses