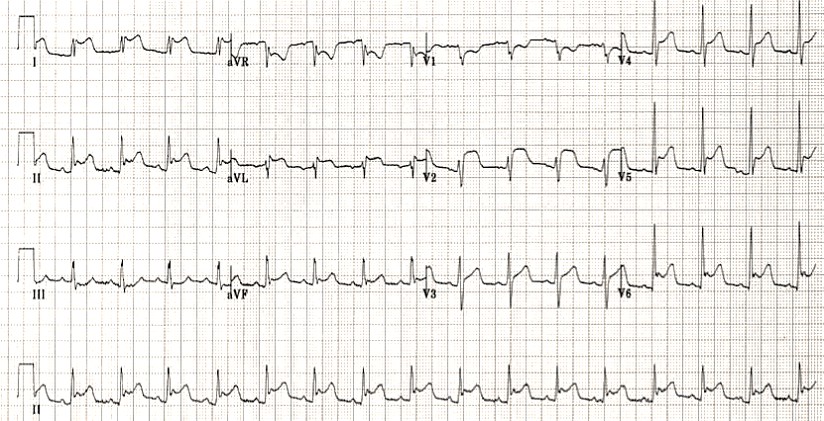
Low electrocardiographic QRS voltage (LQRSV)

Low electrocardiographic QRS voltage (LQRSV) is also called a warning sign.
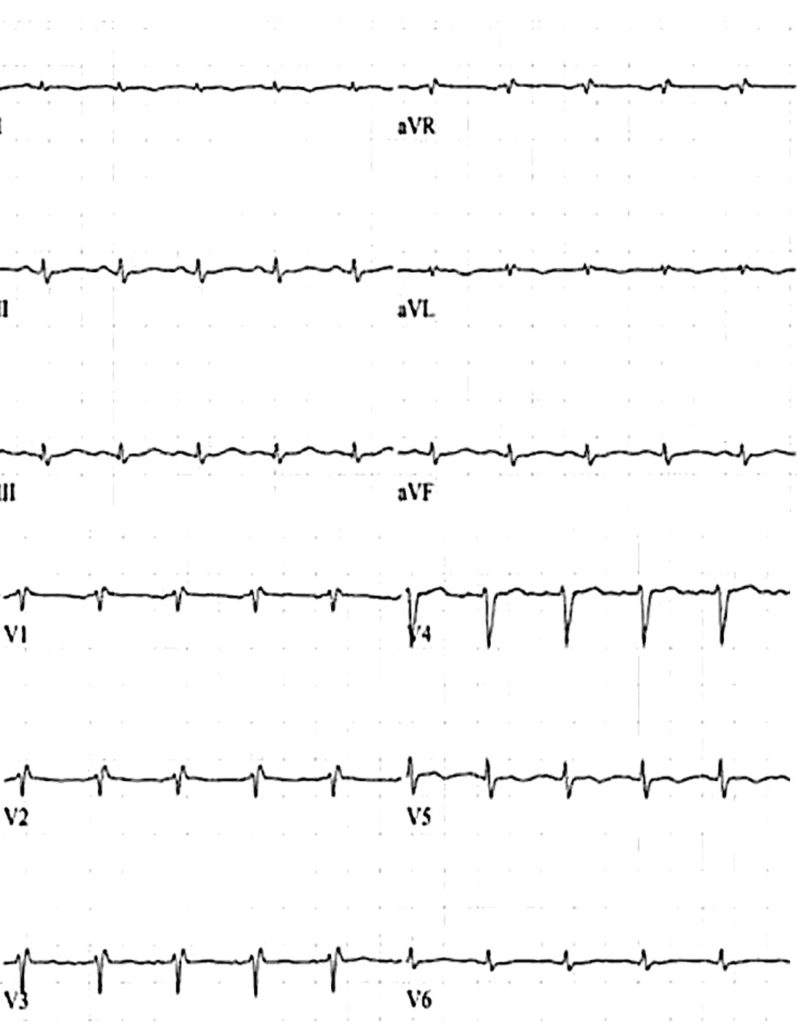
The low-voltage electrocardiogram (ECG) is associated with various cardiac and noncardiac conditions as well as lead wire reversals and other electronic equipment problems. Low-voltage QRS in the ECG creates problems with continuous cardiac monitoring, such as false lethal arrhythmia alarms at central monitoring stations, and general false alarms, thus contributing to alarm fatigue. The low-voltage ECG may be associated with physiological, anatomical features and conditions.
Diagnostic criteria
The QRS has low voltage when:
- The amplitudes of all the QRS complexes in the limb leads are < 5 mm; or
- The amplitudes of all the QRS complexes in the precordial leads are < 10 mm
Low voltage of the QRS amplitude can be defined according to the criteria depicted in Table.
Table: Criteria for ECG voltage measurement
| Limb leads | Precordial leads | Criteria of Carroll et al. |
| ≤0.5 mV in all limb leads | ≤1.0 mV in all precordial leads | SV1 + RV5 or V6 < 1.5 mV |
Mechanisms
Low voltage is produced by:
- The “damping” effect of increased layers of fluid, fat or air between the heart and the recording electrode
- Loss of viable myocardium
- Diffuse infiltration or myxoedematous involvement of the heart
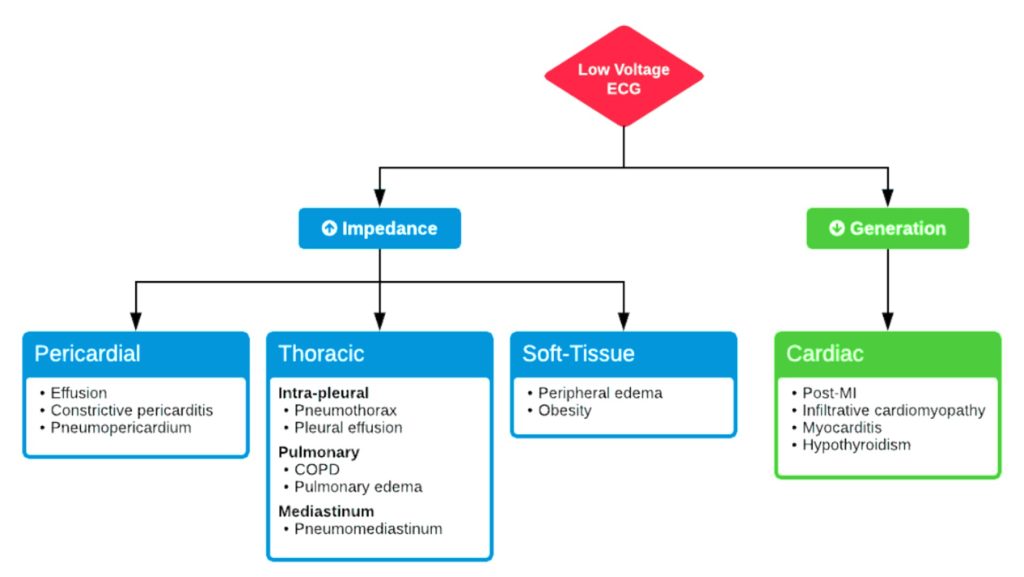
Causes
The most important cause is massive pericardial effusion, which produces a triad of:
- Low voltage
- Tachycardia
- Electrical alternans
Patients with this triad need to be immediately assessed for clinical or echocardiographic evidence of tamponade.
- Fluid: Pericardial effusion; Pleural effusion
- Fat: Obesity
- Air: Chronic obstructive pulmonary disease, or COPD, Emphysema; Pneumothorax; Subcutaneous emphysema
- Infiltrative / Connective Tissue Disorders
- Myxoedema – Severe hypothyroidism
- Infiltrative myocardial diseases — i.e. restrictive cardiomyopathy due to Infiltrative/restrictive diseases such as amyloid cardiomyopathy, sarcoidosis, haemochromatosis
- Constrictive pericarditis
- Scleroderma
- Loss of viable myocardium:
- Massive myocardial damage/infarction
- Previous massive MI
- End-stage dilated cardiomyopathy
Note! If the gain indicated at the left of the ECG is turned down accidentally, the voltage will be falsely low (pseudo low voltage). The indicator should be set to 10 mm amplitude.
Low ECG QRS voltage in limb leads with normal QRS precordial amplitudes, or LQRSV in limb leads with high QRS complexes in the precordial leads with poor R-wave progression (“Goldberger triad”) 3 have been described in patients with dilated cardiomyopathy.
Cardiac causes of LQRSV
- Multiple myocardial infarctions due to cancellations and diminished electromotive force generation;
- LQRSV and QRS notches are seen in conjunction with severe post–myocardial infarction dysynergy
- Infiltrative cardiomyopathies (amyloidosis), which occurs despite the marked cardiac hypertrophy or dilatation
- Myocarditis is associated with LQRSV attributed to the ailing myocytes, although extracardiac influences may also contribute to the LQRSV
- Reduction of QRS voltage (not necessarily LQRSV) follows reduction of cardiac volumes due to various pathologies, hemorrhage, or hypovolemia (“Brody effect”).
- Pulmonary congestion and/ or peripheral edema (PERED) may contribute to LQRSV (vide infra).
- Extensive skin burns may lead to hypovolemia and cause LQRSV, although associated hypoalbuminemia may contribute (vide infra).
- An increased hematocrit leads to LQRSV because of the reduction in disparity of electrical resistivity of the intracardiac blood mass and the surrounding myocardium, which attenuates the radial depolarization forces, as per the Brody effect.
Pericardial causes of LQRSV
The mechanism purported to be that of a short-circuiting of the heart’s potentials as they are transmitted to the body surface;
- the intrapericardial pressure, like in tamponade, as the primary reason, along with the inflammation. However, the delays in recovery of LQRSV after pericardiocentesis or alleviation of tamponade suggest that the effects on the ECG in pericarditis/pericardial effusion/tamponade are multifactorial.
- Electrical alternans, in the presence of a large pericardial effusion, often with impending tamponade, is attributed to a swinging motion of the heart, with a 2-beat or varying periodicity.
- Thickening of the pericardium in constrictive pericarditis is associated with LQRSV20; however, pericardiectomy only partially restores the QRS amplitude, suggesting that the underlying myocardium is contributing to the LQRSV.
Extracardiac causes of LQRSV
The pathology of the organs and tissues surrounding the heart impacts the transfer of the heart’s potential to the body surface with resultant LQRSV.
Patients with chronic obstructive lung disease may show LQRSV, particularly in the limb leads, because of an increased heart/chest wall distance from lung hyperinflation, which, if not offset, would be expected to augment QRS potentials by way of an increased electrical impedance.
Pneumopericardium, pneumomediastinum, and pneumothorax, particularly left-sided, are associated with LQRSV. Pulmonary edema and bronchopulmonary “lavage” result in LQRSV because of decreased lung impedance by way of increased water content. Pneumonia, with extensive pulmonary infiltrates, and adult respiratory distress syndrome (“wet lung”) are expected to lead to similar ECG findings, although nothing to this effect has been described heretofore. Pleural effusion, particularly left-sided and in the absence of congestive heart failure, causes LQRSV with an inverse relationship between the extent of the effusion and the amplitude of QRS complexes. Extensive subcutaneous emphysema, with retro pneumoperitoneum and mediastinal emphysema, is associated with LQRSV. Change in the position of the heart and its relationship with the chest wall, and certain ECG leads must be influential in LQRSV in some of the above-discussed conditions. Obesity is linked to reversible LQRSV.
Although LQRSV is rarely found in hypothyroidism in the absence of pericardial effusion, it has been suggested that hypothyroidism per se contributes to LQRSV.
Sepsis, certain drugs (eg, nonsteroidal anti-inflammatory drugs and the antidiabetic thiazolidinediones [unpublished data]), cor pulmonale, perioperative fluid load administration, even in the presence of normal left ventricular function, chronic renal failure, particularly during the predialytic state, congestive heart failure, hepatic cirrhosis, and numerous other conditions result in reversible LQRSV.
Hypoalbuminemia and resultant PERED must be the mechanism of LQRSV noted invariably in children with kwashiorkor, rather than “myocardial atrophy,” although low cardiac volumes and decreased cardiac thickness and mass may contribute to the LQRSV. Peripheral edema also decreases the amplitude of P waves and T waves, and the duration of P waves, QRS complexes, and QT intervals; such changes have enormous clinical implications for the diagnosis, management, and follow-up of patients with broad categories of cardiac and noncardiac diseases and, in addition, impact the signal-averaged electrocardiogram and T-wave alternans, with major consequences on the reproducibility and clinical relevance of such measurements.
Causes of low voltage based on the mechanism:
| Cardiac causes of LQRSV | – Alcoholic cardiomyopathy – Amyloidosis – Anasarca – Dilated cardiomyopathy – Beriberi heart disease – Cardiomyopathy – Chagas disease – Diffuse coronary artery disease – Congestive heart failure – Constrictive pericarditis – Dilated cardiomyopathy – Generalized edema and pedal edema – Ischemic cardiomyopathy – Prior myocardial infarction – Myocarditis – Rejection after heart transplantation – Restrictive cardiomyopathy -Scleroderma |
| Pericardial causes of LQRSV | – Tamponade |
| Extracardiac causes of LQRSV | – COPD – Emphysema – Myxedema – Obesity – Hemochromatosis – Hypothermia – Hypothyroidism – Pericardial effusion – Pneumothorax – Sarcoidosis |
| Technical error | – A technical error in the settings on the ECG apparatus (sensitivity should be at 10 mm/mV) |
Previously Andrew O. Usoro et al. reported that the mortality rate in individuals with LQRSV was almost twice that in those without LQRSV (51.1 vs 23.5 events per 1,000 person-years, p <0.01).
Register on our website right now to have access to more learning materials!
Subscribe to our pages:
Literarure:
- Cyrille NB, Goldsmith J, Alvarez J, Maurer MS. Prevalence and prognostic significance of low QRS voltage among the three main types of cardiac amyloidosis. The American journal of cardiology. 2014;114(7):1089-1093.
- Mussinelli R, Salinaro F, Alogna A, Boldrini M, Raimondi A, Musca F, Palladini G, Merlini G, Perlini S. Diagnostic and prognostic value of low QRS voltages in cardiac AL amyloidosis. Ann Noninvasive Electrocardiol. 2013;18(3):271-280.
- Carroll JD, Gaasch WH, McAdam KP. Amyloid cardiomyopathy: characterization by a distinctive voltage/mass relation. The American journal of cardiology. 1982;49(1):9-13.
- Madias JE. Low QRS voltage and its causes. J Electrocardiol. 2008;41(6):498–500. doi:10.1016/j.jelectrocard.2008.06.021.
- Andrew O. Usoro, Natalie Bradford, Amit J. Shah, Elsayed Z. Soliman. Risk of Mortality in Individuals With Low QRS Voltage and Free of Cardiovascular Disease. Arrhythmias and conduction disturbances. 2014; Vol. 113(9): P.1514-1517. DOI:https://doi.org/10.1016/j.amjcard.2014.02.006
- Kamath SA, Meo Neto Jde P, Canham RM, Uddin F, Toto KH, Nelson LL, Kaiser PA, de Lemos JA, Drazner MH. Low voltage on the electrocardiogram is a marker of disease severity and a risk factor for adverse outcomes in patients with heart failure due to systolic dysfunction. Am Heart J. 2006 Aug;152(2):355-61. doi: 10.1016/j.ahj.2005.12.021. PMID: 16875922. https://pubmed.ncbi.nlm.nih.gov/16875922/
- Hannibal Gerard B. Interpretation of the Low-Voltage ECG. AACN Adv Crit Care (2014) 25 (1): 64–68.https://doi.org/10.4037/NCI.0000000000000001
Emergency care of a patient with chest pain – Acute Coronary Syndrome with ST-segment elevation and equivalents /OSCE guide
Opening the consultationWash your hands and don PPE if appropriateIntroduce yourself to the patient including…
Stephen Smith’s formula for Subtle Anterior STEMI online Calculator
Differentiates normal variant ST elevation (benign early repolarization) from anterior STEMI, more sensitive than 3-variable…
Corrected QT Interval Bazett’s formula online calculator
Bazett’s formula The formula should be used in patients with a heart rate of 60…
South Africa Flag sign on ECG
South Africa Flag sign on the ECG – a specific ECG pattern that occurs during…
Aslanger’s Pattern
Introduction The fourth universal definition of myocardial infarction (MI) defines ST-segment elevation MI (STEMI) and…
Two masks of the same syndrome: Wellens syndrome
Early reperfusion therapy is a gold standard strategy in the management of patients with acute…